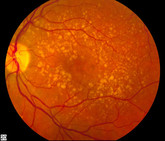
On 23 April 2026, the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) adopted positive opinions, recommending the granting of a marketing authorization for I...
More > 0
0

On 23 April 2026, the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) adopted positive opinions, recommending the granting of a marketing authorization for I...
More >