Bysumlog (insulin lispro), Dazparda (insulin aspart), and Fubelv (etanercept) receive positive CHMP opinions
On 26 February 2026, the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) adopted positive opinions, recommending the granting of a marketing authorization for three biosimilar medicines: Bysumlog (insulin lispro), Dazparda (insulin aspart), and Fubelv (etanercept).
Insulin biosimilars
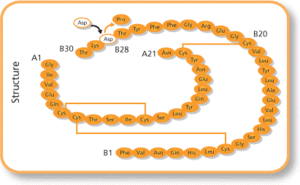
The applicant for Bysumlog and Dazparda is Gan & Lee Pharmaceuticals Europe. Its headquarters are based in Beijing, Cihna. Both insulin lispro and insulin aspart are used in treatment of diabetes. They are fast-acting insulin analogues which have a more rapid onset of action than human soluble insulin. Both products will be available as a 100 unit/mL solution for injection in a pre-filled pen. The originators are Eli Lilly’s Humalog (insulin lispro) and Novo Nordisk’s NovoLog (insulin aspart) [1].
Bysumlog (Insulin Lispro)
The biosimilarity application for Bysumlog was supported by a randomized, double-blind Phase 1 clinical study conducted in Europe. This study evaluated the pharmacokinetics (PK) and pharmacodynamics (PD) of Gan & Lee's insulin lispro compared to the reference product, Humalog. The results demonstrated that Bysumlog is bioequivalent to Humalog in terms of PK/PD profiles, with a comparable safety and tolerability profile . Specifically, the product is recommended for the treatment of diabetes mellitus in adults and children.
Dazparda (Insulin Aspart)
Similarly, the approval for Dazparda was based on a randomized, double-blind Phase I PK/PD comparative study in Europe. The study confirmed the biosimilarity between Dazparda and the reference products NovoLog/NovoRapid. The data showed no clinically meaningful differences in efficacy, safety, or immunogenicity between the biosimilar and the originator . The CHMP recommended Dazparda for the treatment of diabetes mellitus in adults, adolescents, and children aged 1 year and older.
Gan & Lee entered into development, commercialization and license agreement with Sandoz for three insulin biosimilars (Insulin Glargine, Insulin Lispro and Insulin Aspart). Under this agreement, following regulatory approval of the products, Sandoz has the right to commercialize the three biosimilars from Gan & Lee upon approval in several regions, including Europe, and other key territories, while Gan & Lee will be responsible for manufacturing and supply.
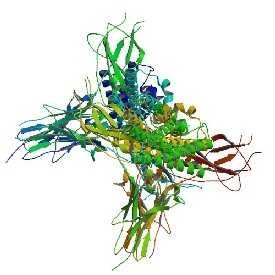
Fubelv (Etanercept)
Etanercept is a fusion protein, which preferentially binds to tumour necrosis factor alpha (TNF-α) and thereby inhibits its biological activity [3].
The applicant for Fubelv is Biosimilar Collaborations Ireland Limited. The positive opinion for Fubelv was based on comprehensive data demonstrating comparable quality, safety, and efficacy to the reference medicine Enbrel, as required for biosimilar approval by the EMA.
The recommended indications for Fubelv include the treatment of several autoimmune and inflammatory conditions such as rheumatoid arthritis, juvenile idiopathic arthritis, psoriatic arthritis, axial spondyloarthritis (including ankylosing spondylitis), and plaque psoriasis (in adults and children from 6 years of age) .
Fubelv will be available as 25 mg and 50 mg solutions for injection. The originator is Amgen’s Enbrel.
Regulatory Context
In January 2026, six new medicines were recommended for approval by the EMA, with no biosimilars approved during that month.
Related articles
EMA recommends approval for pertuzumab and tocilizumab biosimilars
EMA recommends approval for teriparatide biosimilar Zandoriah
EMA recommends approval for insulin glargine biosimilar Ondibta and denosumab biosimilar Osqay
References
1. GaBI Online - Generics and Biosimilars Initiative. Biosimilars of insulin aspart [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2026 Apr 16]. Available from: www.gabionline.net/biosimilars/general/biosimilars-of-insulin-aspart
2. GaBI Online - Generics and Biosimilars Initiative. Biosimilars of insulin etanercept [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2026 Apr 16]. Available from: www.gabionline.net/biosimilars/general/biosimilars-of-etanercept
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2026 Pro Pharma Communications International. All Rights Reserved.








 0
0







Post your comment