Biosimilars
Research on biosimilars in anaemia and diabetes
Period: September to December 2012
The use of biological medicines has been life-changing for many patients suffering with anaemia and diabetes. With the expiration of patents on these biologicals the possibility of patients gaining access to lower cost biosimilar alternatives to these extremely effective, but costly, medicines becomes a real possibility.
Biosimilars in rheumatology
Patents on key biological medicines used in rheumatology will expire soon. The European Medicines Agency’s (EMA’s) finalized guidelines on biosimilar monoclonal antibodies (mAbs) came into effect on 1 December 2012. Both of these facts mean that rheumatologists can expect to be exposed to biosimilars of the medicines they routinely use in the near future.
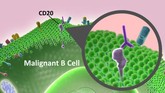
Sandoz’s biosimilar rituximab on track
The development of a biosimilar version of Rituxan (rituximab) is proceeding as planned, Sandoz’s Global Head, Mr Jeff George told Bloomberg.
How are biosimilars special
Despite biosimilars being around in the EU since 2006 physicians are still often reluctant to prescribe them. Members and experts of the Working Party on Similar Biologic Medicinal Products of the European Medicines Agency (EMA) highlight what physicians need to know to make informed and appropriate treatment choices for their patients [1].
When will the US biosimilars pathway be used
FDA Commissioner Margaret Hamburg presented the latest statistics on the US biosimilar pathway during a speech at the Massachusetts Biotechnology Council’s (MassBio) Annual Meeting on 15 March 2013.
Greece says no to automatic substitution of biologicals
Greece’s Medicines Agency (National Organization for Medicines, Ethnikos Organismos Farmakon, EOF) released a document on 13 March 2013 recommending against automatic substitution/interchangeability of reference biologicals and their biosimilars.
Pharma companies sue EMA to block release of data
US-based biotech companies AbbVie and InterMune have both sued EMA in a bid to block publication of clinical trial data for their drugs.
Research on biosimilars in rheumatology
Period: September to December 2012
Over the past decade, the availability of targeted biological therapies has revolutionized the treatment of rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis. However, the significant cost of these biologicals is often prohibitive and limits universal access to these effective therapeutic agents. Whereas generic drug equivalents are commercially available for many small-molecule medications, such lower cost alternatives to targeted biological therapies are not yet available in the US or the EU. The first biological therapeutics in rheumatology is now approaching patent expiration and biosimilars are now in randomized controlled trials. This means that cheaper biosimilars for the treatment of rheumatic diseases are likely to enter the market in the near future, increasing patient access to these life-changing treatments.
Japan approves second biosimilar G-CSF
Japan has granted regulatory approval to a second biosimilar granulocyte colony-stimulating factor (G-CSF).
XBiotech moves into biosimilars
US-based biotech company XBiotech announced on 26 February 2013 that the company would be expanding its portfolio to include biosimilars.