Biosimilars/News
Amgen to start phase III trial for biosimilar adalimumab
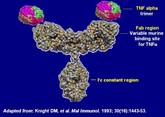
Biotechnology giant Amgen is to start a phase III clinical trial for a biosimilar version of adalimumab in patients suffering from severe rheumatoid arthritis according to the EU Clinical Trials Register.
EMA approves biosimilar follitropin alfa and somatropin
The European Medicines Agency’s (EMA’s) Committee for Medicinal Products for Human Use (CHMP) announced on 31 July 2013 that it had recommended granting of marketing authorization for a biosimilar follitropin alfa. While on 9 September 2013, the agency announced the approval of a new somatropin biosimilar.
Failed biosimilars company to be sold
The assets of biosimilars developer Elona Biotechnologies (Elona) will be auctioned on 27 September 2013 unless sold before 6 September 2013.
Celltrion applies for biosimilar infliximab approval in Japan
On 11 September 2013, South Korean biotechnology company Celltrion announced that it had filed for approval of Remsima, its biosimilar infliximab monoclonal antibody, with Japan’s Ministry of Health, Labour and Welfare (MHLW).
Baxter and Coherus to collaborate on biosimilars
Baxter International (Baxter) has made another agreement for biosimilars, this time with fledgling biotech company Coherus Biosciences (Coherus).
Roche abandons Herceptin patent in India
Swiss-based pharma giant Roche has decided not to pursue its patent for its blockbuster cancer drug Herceptin (trastuzumab) in India, opening the market to biosimilars.
Indian firms chasing ‘similar biologic’ Herceptin
Following the decision by Swiss-based pharma giant Roche not to pursue its patent for Herceptin (trastuzumab) in India [1] it seems that a whole host of Indian firms are lining up to get their ‘similar biologic’ of the blockbuster cancer drug to market.
Apotex breaking into the North American and European G-CSF markets
A new biosimilar filgrastim is poised to enter the important granulocyte colony-stimulating factor (G-CSF) market following a positive opinion from the European Medicines Agency’s (EMA’s) Committee for Medicinal Products for Human Use (CHMP).
EMA approval for Teva’s long-acting recombinant G-CSF
The European Medicines Agency (EMA) has granted marketing authorization for a new long-acting filgrastim – Lonquex (XM22 lipegfilgrastim) – to prevent chemotherapy-induced neutropenia in cancer patients.
AstraZeneca pursuing biosimilars with Celltrion bid
Pharma giant AstraZeneca is rumoured to be ready to join the race for biosimilars with a bid for South Korean biotechnology company Celltrion.