Generics/News
FDA approves generic teriparatide and levetiracetam
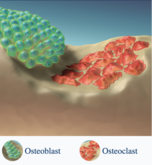
Two generic versions of osteoporosis treatment, Forteo/Forsteo (teriparatide injection), developed by Teva and Ambio, were approved by the US Food and Drug administration (FDA) in November and December 2023. Although teriparatide is a biological, the new products have been produced synthetically and are therefore generic versions and not biosimilars, of the originator. In addition, Strides Pharma has received approval for its generic seizure medication, levetiracetam.
US generics launch and approval for Dr Reddy’s and Lupin
On 7 September 2022, Dr Reddy’s Laboratories (Dr Reddy’s) announced the US launch of their generic lenalidomide capsules. And in July 2022, Lupin announced that its generic azilsartan medoxomil tablets had been approved by the US Food and Drug Administration (FDA).
Five Chinese companies join UN’s MPP for Covid-19 medicines
Five pharmaceutical companies in China have signed up to the United Nation’s (UN) Medicine Patent Pool (MPP) which will allow them to produce generic versions of Pfizer's oral COVID-19 treatment, Paxlovid (nirmatrelvir/ritonavir), for distribution in 95 low- and middle-income countries, approximately 53 per cent of the world's population.
South Korean companies to make generic Bridion and COVID-19 drugs
In South Korea, 19 pharmaceutical companies have obtained marketing approval for generic drugs of MSD’s Bridion (sugammadex). In addition, Korean pharma companies Celltrion, Dongbang and Hanmi Pharmaceutical, will be supplying generic COVID-19 drugs to middle- and low-income countries.
Revlimid (lenalidomide) generics launch across Europe
In late February 2022, a number of generics of Revlimid (lenalidomide) were launched in Europe. These will now rival Bristol Meyers Squibb’s blockbuster oncology medicine.
Cancer and diabetes generic treatments receive positive opinion from EMA’s CHMP
The European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) announced on 28 January 2022 that three generic medicines had received a positive opinion from the committee.
FDA approves first Restasis generic
On 2 February 2022, the US Food and Drug Administration (FDA) approved the first generic of Restasis (cyclosporine ophthalmic emulsion).
FDA approves schizophrenia and anti-allergy drugs
The US Food and Drug Administration (FDA) has granted tentative approvals to Zydus Cadila and Morepen Laboratories for treatments for schizophrenia and allergy symptoms, respectively.
Pfizer to produce generic COVID-19 pill in eight countries of Latin America
US-based pharmaceutical company Pfizer announced a licensing agreement to allow other manufacturers to produce generic versions of its COVID-19 pill Paxlovid (PF-07321332*) in order to generate more affordable access to developing countries.
First eslicarbazepine epilepsy generic to be approved in China
In China, the Yangtze River Pharmaceutical Group submitted the first marketing application of a class 3 generic drug, eslicarbazepine acetate, for use to treat patients with epilepsy, on 15 October 2021.