Canada’s drug regulator, Health Canada, has approved the rituximab biosimilar Riabni for the treatment of rheumatoid arthritis as well as chronic lymphocytic leukaemia and non-Hodgkin’s lymphoma.
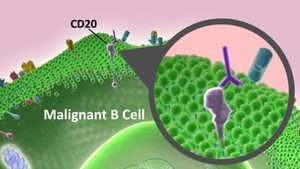
The drug is a biosimilar of Roche’s Rituxan (rituximab). Rituximab is a chimeric monoclonal antibody against the protein CD20, which is primarily found on the surface of immune system B cells. Rituximab destroys B cells and is therefore used to treat diseases that are characterized by excessive number of B cells, overactive B cells or dysfunctional B cells. This includes many lymphomas, leukaemias, transplant rejection and autoimmune disorders.
Amgen’s Riabni (ABP 798) was approved on 11 March 2021 as a solution for intravenous injection at a dose of 10 mg/mL.
This latest approval brings the total rituximab biosimilars approved in Canada up to four.
Teva Canada Innovation, a subsidiary of Teva Pharmaceutical Industries, had its rituximab biosimilar, Truxima (CT P10), approved by Health Canada in April 2019 [1]. This is the first rituximab biosimilar approved in Canada. This was followed in April 2020 by Sandoz’s Riximyo and in May 2020 by Pfizer’s Ruxience.
The drugs are currently under consideration for public reimbursement by the pan-Canadian Pharmaceutical Alliance for public reimbursement on federal, provincial and territorial drug formularies.
Related article
Biosimilars of rituximab
Reference
1. GaBI Online - Generics and Biosimilars Initiative. Biosimilars approved in Canada [www.gabionline.net]. Mol, Belgium: Pro Pharma Communications International; [cited 2021 Jul 9]. Available from: www.gabionline.net/biosimilars/general/biosimilars-approved-in-canada
| LATIN AMERICAN FORUM The brand-new section the ‘Latin American Forum’ on GaBI has been launched. The objective of this new section is to provide you with all the latest news and updates on developments of generic and biosimilar medicines in Latin America in Spanish. View this week’s headline article: Biosimilares en Japón: una perspectiva general Browse the news in the Latin American Forum! Register to receive the GaBI Latin American Forum newsletter. Inform colleagues and friends of this new initiative. LATIN AMERICAN FORUM Se ha lanzado la nueva sección del ‘Foro Latinoamericano’ sobre GaBI. El objetivo de esta nueva sección es brindarle las últimas noticias y actualizaciones sobre desarrollos de medicamentos genéricos y biosimilares en América Latina en español. Vea el artículo principal de esta semana: Biosimilares en Japón: una perspectiva general Explore las noticias en el Foro Latinoamericano! Regístrese para recibir el boletín informativo GaBI Latin American Forum. Informe a colegas y amigos sobre esta nueva iniciativa.
|
Permission granted to reproduce for personal and non-commercial use only. All other reproduction, copy or reprinting of all or part of any ‘Content’ found on this website is strictly prohibited without the prior consent of the publisher. Contact the publisher to obtain permission before redistributing.
Copyright – Unless otherwise stated all contents of this website are © 2021 Pro Pharma Communications International. All Rights Reserved.








 0
0










Post your comment